is the combustion product gas from a
fireplace, oven, furnace, boiler or steam generator that exits to the
atmosphere via a ''flue'' which may be a pipe, channel or chimney. The
flue is most commonly referred to as a "flue gas stack" by engineers or a
"smokestack" by lay people.
Flue gases are produced when natural gas, fuel oil, coal, wood or any
other fuel is combusted in an industrial furnace, a steam generator in a
fossil fuel power plant or other combustion sources.
)
and water vapor as well as nitrogen and excess oxygen remaining from
the intake combustion air. It may also contain a small percentage of air
pollutants such as particulate matter, carbon monoxide, nitrogen
oxides, sulfur oxides and mercury. Typically, more than two-thirds of
the flue gas is nitrogen.
The table below provides the amount of flue gas (on a dry basis as well
as a wet basis) generated by burning a typical fuel gas, fuel oil or
coal. The flue gas amounts were obtained by stoichiometric calculations
using the indicated typical excess combustion air percentages:
Given the amount of gas, oil or coal fuel burned in a combustion device,
then the flue gas generation data (i.e., m³/GJ of fuel) in the above
table provides a basis for estimating the amount of flue gas generated.
At power plants, flue gas is often treated with a series of chemical
processes and scrubbers, which remove pollutants. Electrostatic
precipitators or fabric filters remove particulate matter and flue gas
desulfurization removes the sulfur dioxide (SO2) produced by burning fossil fuels, particularly coal.
Nitrogen oxides emissions are reduced either by modifications to the
combustion process to prevent their formation, or by catalytic reaction
with ammonia or urea. In either case, the aim is to produce nitrogen
gas, rather than nitrogen oxides.
In the United States, there is also a rapid deployment of technologies
to remove mercury from flue gas - typically by adsorption on sorbents or
by capture in inert solids as part of the flue gas desulfurization
product.
Technologies for the removal and capture of carbon dioxide from flue
gases are now under active research and development as a means of
reducing the emissions of so-called ''greenhouse gas''.
ANALYSIS OF FLUE GASES
The object of a flue gas analysis is the determination of the
completeness of the combustion of the carbon in the fuel, and the amount
and distribution of the heat losses due to incomplete combustion. The
quantities actually determined by an analysis are the relative
proportions by volume, of carbon dioxide (CO
2), oxygen (O), and
carbon monoxide (CO), the determinations being made in this order.
The variations of the percentages of these gases in an analysis is best
illustrated in the consideration of the complete combustion of pure
carbon, a pound of which requires 2.67 pounds of oxygen,
[28] or 32 cubic
feet at 60 degrees Fahrenheit. The gaseous product of such combustion
will occupy, when cooled, the same volume as the oxygen, namely, 32
cubic feet. The air supplied for the combustion is made up of 20.91 per
cent oxygen and 79.09 per cent nitrogen by volume. The carbon united
with the oxygen in the form of carbon dioxide will have the same volume
as the oxygen in the air originally supplied. The volume of the nitrogen
when cooled will be the same as in the air supplied, as it undergoes no
change. Hence for complete combustion of one pound of carbon, where no
excess of air is supplied, an analysis of the products of combustion
will show the following percentages by volume:
| Actual Volume
for One Pound Carbon
Cubic Feet | | Per Cent
by Volume |
|---|
| Carbon Dioxide | 32 | = | 20.91 |
| Oxygen | 0 | = | 0.00 |
| Nitrogen | 121 | = | 79.09 |
| –––––– | | ––––––––––– |
| Air required for one pound Carbon | 153 | = | 100.00 |
For 50 per cent excess air the volume will be as follows:
| 153 × 1½ = 229.5 cubic feet of air per pound of carbon. |
| Actual Volume
for One Pound Carbon
Cubic Feet | | Per Cent
by Volume | |
|---|
| Carbon Dioxide | 32 | = | 13.91 | } | = | 20.91 per cent |
| Oxygen | 16 | = | 7.00 |
| Nitrogen | 181.5 | = | 79.09 | |
| –––––––––– | | ––––––––––– | |
| Air required for one pound Carbon | 229.5 | = | 100.00 | |
For 100 per cent excess air the volume will be as follows:
| 153 × 2 = 306 cubic feet of air per pound of carbon. |
| Actual Volume
for One Pound Carbon
Cubic Feet | | Per Cent
by Volume | |
|---|
| Carbon Dioxide | 32 | = | 10.45 | } | = | 20.91 per cent |
| Oxygen | 32 | = | 10.45 |
| Nitrogen | 242 | = | 79.09 | |
| –––––– | | ––––––––––– | |
| Air required for one pound Carbon | 306 | = | 100.00 | |
In each case the volume of oxygen which combines with the carbon is
equal to (cubic feet of air × 20.91 per cent)—32 cubic feet.
It will be seen that no matter what the excess of air supplied, the
actual amount of carbon dioxide per pound of carbon remains the same,
while the percentage by volume decreases as the excess of air increases.
The actual volume of oxygen and the percentage by volume increases with
the excess of air, and the percentage of oxygen is, therefore, an
indication of the amount of excess air. In each case the sum of the
percentages of CO
2 and O is the same, 20.9. Although the volume of
nitrogen increases with the excess of air, its percentage by volume
remains the same as it undergoes no change while combustion takes place;
its percentage for any amount of air excess, therefore, will be the same
after combustion as before, if cooled to the same temperature. It must
be borne in mind that the above conditions hold only for the perfect
combustion of a pound of pure carbon.
Carbon monoxide (CO) produced by the imperfect combustion of carbon,
will occupy twice the volume of the oxygen entering into its composition
and will increase the volume of the flue gases over that of the air
supplied for combustion in the proportion of
| 1 to |
| 100 + ½ the per cent CO |
| ––––––––––––––––––––––––––––––––––––––– |
| 100 |
|
When pure carbon is the fuel, the sum of the percentages by volume of
carbon dioxide, oxygen and one-half of the carbon monoxide, must be in
the same ratio to the nitrogen in the flue gases as is the oxygen to the
nitrogen in the air supplied, that is, 20.91 to 79.09. When burning
coal, however, the percentage of nitrogen is obtained by subtracting the
sum of the percentages by volume of the other gases from 100. Thus if an
analysis shows 12.5 per cent CO
2, 6.5 per cent O, and 0.6 per cent
CO, the percentage of nitrogen which ordinarily is the only other
constituent of the gas which need be considered, is found as follows:
| 100 - (12.5 + 6.5 + 0.6) = 80.4 per cent. |
The action of the hydrogen in the volatile constituents of the fuel is
to increase the apparent percentage of the nitrogen in the flue gases.
This is due to the fact that the water vapor formed by the combustion of
the hydrogen will condense at a temperature at which the analysis is
made, while the nitrogen which accompanied the oxygen with which the
hydrogen originally combined maintains its gaseous form and passes into
the sampling apparatus with the other gases. For this reason coals
containing high percentages of volatile matter will produce a larger
quantity of water vapor, and thus increase the apparent percentage of
nitrogen.
Air Required and Supplied—When the ultimate analysis of a fuel is
known, the air required for complete combustion with no excess can be
found as shown in the chapter on combustion, or from the following
approximate formula:
| Pounds of air required per pound of fuel | = | 34.56 | ( |
| + | ( | H | - |
| ) | + |
| ) | [29] (11) |
where C, H and O equal the percentage by weight of carbon, hydrogen and
oxygen in the fuel divided by 100.
When the flue gas analysis is known, the total, amount of air supplied
is:
| Pounds of air supplied per pound of fuel | = | 3.036 | ( |
| N |
| ––––––––––––––––– |
| CO2 + CO |
| ) | × | C | [30] | (12) |
where N, CO
2 and CO are the percentages by volume of nitrogen, carbon
dioxide and carbon monoxide in the flue gases, and C the percentage by
weight of carbon which is burned from the fuel and passes up the stack
as flue gas. This percentage of C which is burned must be distinguished
from the percentage of C as found by an ultimate analysis of the fuel.
To find the percentage of C which is burned, deduct from the total
percentage of carbon as found in the ultimate analysis, the percentage
of unconsumed carbon found in the ash. This latter quantity is the
difference between the percentage of ash found by an analysis and that
as determined by a boiler test. It is usually assumed that the entire
combustible element in the ash is carbon, which assumption is
practically correct. Thus if the ash in a boiler test were 16 per cent
and by an analysis contained 25 per cent of carbon, the percentage of
unconsumed carbon would be 16 × .25 = 4 per cent of the total coal
burned. If the coal contained by ultimate analysis 80 per cent of carbon
the percentage burned, and of which the products of combustion pass up
the chimney would be 80 - 4 = 76 per cent, which is the correct figure
to use in calculating the total amount of air supplied by formula (
12).
The weight of flue gases resulting from the combustion of a pound of dry
coal will be the sum of the weights of the air per pound of coal and the
combustible per pound of coal, the latter being equal to one minus the
percentage of ash as found in the boiler test. The weight of flue gases
per pound of dry fuel may, however, be computed directly from the
analyses, as shown later, and the direct computation is that ordinarily
used.
The ratio of the air actually supplied per pound of fuel to that
theoretically required to burn it is:
| 3.036 | ( |
| N |
| ––––––––––––––––– |
| CO2 + CO |
| ) | × | C |
| (13) |
| ––––––––––––––––––––––––––––––––––––––––––––– |
|
|
|
in which the letters have the same significance as in formulae (
11) and
(
12).
The ratio of the air supplied per pound of combustible to the amount
theoretically required is:
| N |
| –––––––––––––––––––––––––––––––––– |
| N - 3.782(O - ½CO) |
| (14) |
which is derived as follows:
The N in the flue gas is the content of nitrogen in the whole amount of
air supplied. The oxygen in the flue gas is that contained in the air
supplied and which was not utilized in combustion. This oxygen was
accompanied by 3.782 times its volume of nitrogen. The total amount of
excess oxygen in the flue gases is (O - ½CO); hence N - 3.782(O - ½CO)
represents the nitrogen content in the air actually required for
combustion and N ÷ (N - 3.782[O - ½CO]) is the
[Pg 158] ratio of the air supplied
to that required. This ratio minus one will be the proportion of excess
air.
| The heat lost in the flue gases is L = 0.24 W (T - t) (15) |
|
| Where | L | = | B. t. u. lost per pound of fuel, |
| W | = | weight of flue gases in pounds per pound of dry coal, |
| T | = | temperature of flue gases, |
| t | = | temperature of atmosphere, |
| 0.24 | = | specific heat of the flue gases. |
The weight of flue gases, W, per pound of carbon can be computed
directly from the flue gas analysis from the formula:
| 11 CO2 + 8 O + 7 (CO + N) |
| ––––––––––––––––––––––––––––––––––––––––––––– |
| 3 (CO2 + CO) |
| (16) |
where CO
2, O, CO, and N are the percentages by volume as determined
by the flue gas analysis of carbon dioxide, oxygen, carbon monoxide and
nitrogen.
The weight of flue gas per pound of dry coal will be the weight
determined by this formula multiplied by the percentage of carbon in the
coal from an ultimate analysis.
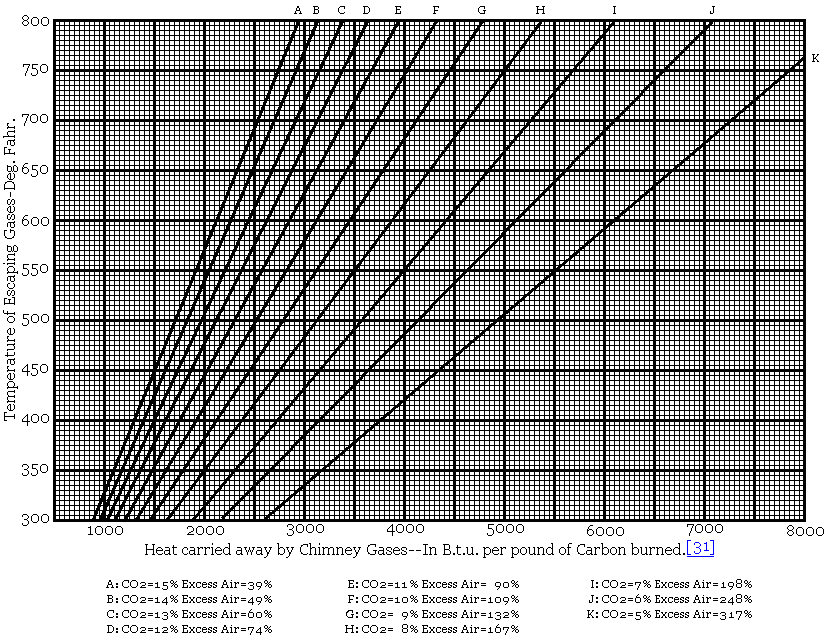
Fig. 20. Loss Due to Heat Carried Away by Chimney Gases for Varying
Percentages of Carbon Dioxide.
Based on Boiler Room Temperature = 80
Degrees Fahrenheit.
Nitrogen in Flue Gas = 80.5 Per Cent. Carbon
Monoxide in Flue Gas = 0. Per Cent
Fig. 20 represents graphically the loss due to heat carried away by dry
chimney gases for varying percentages of CO
2, and different
temperatures of exit gases.
[Pg 159]
The heat lost, due to the fact that the carbon in the fuel is not
completely burned and carbon monoxide is present in the flue gases, in
B. t. u. per pound of fuel burned is:
| L' | = | 10,150 | × | ( |
| CO |
| ––––––––––––––––– |
| CO + CO2 |
| ) | (17) |
where, as before, CO and CO
2 are the percentages by volume in the
flue gases and C is the proportion by weight of carbon which is burned
and passes up the stack.
Fig. 21 represents graphically the loss due to such carbon in the fuel
as is not completely burned but escapes up the stack in the form of
carbon monoxide.
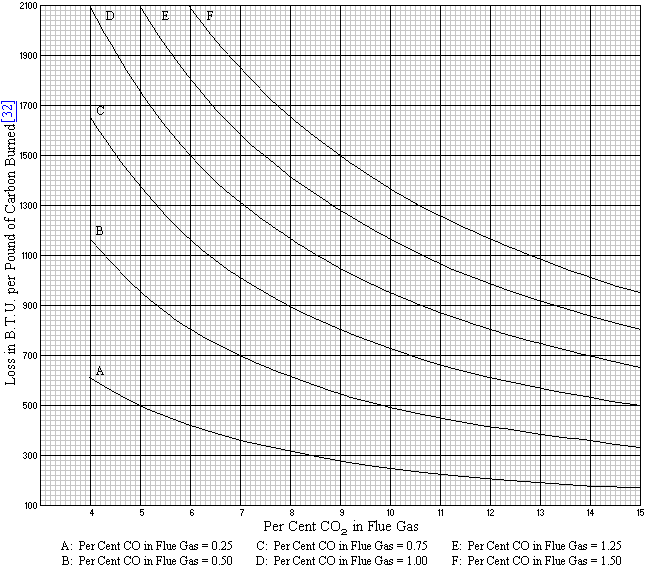
Fig. 21. Loss Due to Unconsumed Carbon Contained in the
CO in the Flue Gases
Apparatus for Flue Gas Analysis—The Orsat apparatus, illustrated in
Fig. 22, is generally used for analyzing flue gases. The burette
A is
graduated in cubic centimeters up to 100, and is surrounded by a water
jacket to prevent any change in temperature from affecting the density
of the gas being analyzed.
For accurate work it is advisable to use four pipettes,
B,
C,
D,
E, the
first containing a solution of caustic potash for the absorption of
carbon dioxide, the second an alkaline solution of pyrogallol for the
absorption of oxygen, and the remaining two an acid solution of cuprous
chloride for absorbing the carbon monoxide. Each pipette contains a
number of glass tubes, to which some of the solution clings, thus
facilitating
[Pg 160] the absorption of the gas. In the pipettes
D and
E, copper
wire is placed in these tubes to re-energize the solution as it becomes
weakened. The rear half of each pipette is fitted with a rubber bag, one
of which is shown at
K, to protect the solution from the action of the
air. The solution in each pipette should be drawn up to the mark on the
capillary tube.
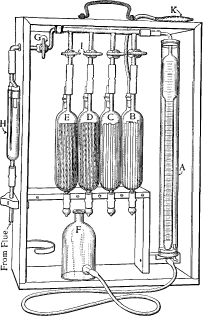
Fig. 22. Orsat Apparatus
The gas is drawn into the burette through the U-tube
H, which is filled
with spun glass, or similar material, to clean the gas. To discharge any
air or gas in the apparatus, the cock
G is opened to the air and the
bottle
F is raised until the water in the burette reaches the 100 cubic
centimeters mark. The cock
G is then turned so as to close the air
opening and allow gas to be drawn through
H, the bottle
F being lowered
for this purpose. The gas is drawn into the burette to a point below the
zero mark, the cock
G then being opened to the air and the excess gas
expelled until the level of the water in
F and in
A are at the zero
mark. This operation is necessary in order to obtain the zero reading at
atmospheric pressure.
The apparatus should be carefully tested for leakage as well as all
connections leading thereto. Simple tests can be made; for example: If
after the cock
G is closed, the bottle
F is placed on top of the frame
for a short time and again brought to the zero mark, the level of the
water in
A is above the zero mark, a leak is indicated.
Before taking a final sample for analysis, the burette
A should be
filled with gas and emptied once or twice, to make sure that all the
apparatus is filled with the new gas. The cock
G is then closed and the
cock
I in the pipette
B is opened and the gas driven over into
B by
raising the bottle
F. The gas is drawn back into
A by lowering
F and
when the solution in
B has reached the mark in the capillary tube, the
cock
I is closed and a reading is taken on the burette, the level of the
water in the bottle
F being brought to the same level as the
water in
A. The operation is repeated until a constant reading is obtained, the
number of cubic centimeters being the percentage of CO
2 in the flue
gases.
The gas is then driven over into the pipette
C and a similar operation
is carried out. The difference between the resulting reading and the
first reading gives the percentage of oxygen in the flue gases.
The next operation is to drive the gas into the pipette
D, the gas being
given a final wash in
E, and then passed into the pipette
C to
neutralize any hydrochloric acid fumes which may have been given off by
the cuprous chloride solution, which, especially if it be old, may give
off such fumes, thus increasing the volume of the gases and making the
reading on the burette less than the true amount.
The process must be carried out in the order named, as the pyrogallol
solution will also absorb carbon dioxide, while the cuprous chloride
solution will also absorb oxygen.
[Pg 161]
As the pressure of the gases in the flue is less than the atmospheric
pressure, they will not of themselves flow through the pipe connecting
the flue to the apparatus. The gas may be drawn into the pipe in the way
already described for filling the apparatus, but this is a tedious
method. For rapid work a rubber bulb aspirator connected to the air
outlet of the cock
G will enable a new supply of gas to be drawn into
the pipe, the apparatus then being filled as already described. Another
form of aspirator draws the gas from the flue in a constant stream, thus
insuring a fresh supply for each sample.
The analysis made by the Orsat apparatus is volumetric; if the analysis
by weight is required, it can be found from the volumetric analysis as
follows:
Multiply the percentages by volume by either the densities or the
molecular weight of each gas, and divide the products by the sum of all
the products; the quotients will be the percentages by weight. For most
work sufficient accuracy is secured by using the even values of the
molecular weights.
The even values of the molecular weights of the gases appearing in an
analysis by an Orsat are:
| Carbon Dioxide | | 44 |
| Carbon Monoxide | | 28 |
| Oxygen | | 32 |
| Nitrogen | | 28 |
Table 33 indicates the method of converting a volumetric flue gas
analysis into an analysis by weight.
TABLE 33
CONVERSION OF A FLUE GAS ANALYSIS BY VOLUME TO ONE BY WEIGHT
| Gas | Analysis by Volume
Per Cent | Molecular Weight | Volume times
Molecular Weight | Analysis by Weight
Per Cent |
|---|
| Carbon Dioxide | CO2 | 12.2 | 12+(2×16) | 536.8 |
|
| Carbon Monoxide | CO | .4 | 12+16 | 11.2 |
|
| Oxygen | O | 6.9 | 2×16 | 220.8 |
|
| Nitrogen | N | 80.5 | 2×14 | 2254.0 |
| 2254.0 |
| ––––––––––– |
| 3022.8 |
| = | 74.6 |
|
| Total | 100.0 | | 3022.8 |
|
|
Application of Formulae and Rules—Pocahontas coal is burned in the
furnace, a partial ultimate analysis being:
| Per Cent |
|---|
| Carbon | 82.1 |
| Hydrogen | 4.25 |
| Oxygen | 2.6 |
| Sulphur | 1.6 |
| Ash | 6.0 |
| B. t. u., per pound dry | 14500 |
[Pg 162]
The flue gas analysis shows:
| Per Cent |
|---|
| CO2 | 10.7 |
| O | 9.0 |
| CO | 0.0 |
| N (by difference) | 80.3 |
Determine: The flue gas analysis by weight (see
Table 33), the amount of
air required for perfect combustion, the actual weight of air per pound
of fuel, the weight of flue gas per pound of coal, the heat lost in the
chimney gases if the temperature of these is 500 degrees Fahrenheit, and
the ratio of the air supplied to that theoretically required.
Solution: The theoretical weight of air required for perfect combustion,
per pound of fuel, from formula (
11) will be,
| W | = | 34.56 |
| = | 10.88 pounds. |
If the amount of carbon which is burned and passes away as flue gas is
80 per cent, which would allow for 2.1 per cent of unburned carbon in
terms of the total weight of dry fuel burned, the weight of dry gas per
pound of carbon burned will be from formula (
16):
| W | = |
| 11 × 10.7 + 8 × 9.0 + 7(0 + 80.3) |
| ––––––––––––––––––––––––––––––––––––––––––––––––––––– |
| 3 (10.7 + 0) |
| = | 23.42 pounds |
and the weight of flue gas per pound of coal burned will be .80 × 23.42
= 18.74 pounds.
The heat lost in the flue gases per pound of coal burned will be from
formula (
15) and the value 18.74 just determined.
Loss = .24 × 18.74 × (500 - 60) = 1979 B. t. u.
The percentage of heat lost in the flue gases will be 1979 ÷ 14500 =
13.6 per cent.
The ratio of air supplied per pound of coal to that theoretically
required will be 18.74 ÷ 10.88 = 1.72 per cent.
The ratio of air supplied per pound of combustible to that required will
be from formula (
14):
| .803
|
| ––––––––––––––––––––––––––––––––––––––– |
| .803 - 3.782(.09 - ½ × 0) |
| = | 1.73 |
The ratio based on combustible will be greater than the ratio based on
fuel if there is unconsumed carbon in the ash.
Unreliability of CO2 Readings Taken Alone—It is generally assumed
that high CO
2 readings are indicative of good combustion and hence of
high efficiency. This is true only in the sense that such high readings
do indicate the small amount of excess air that usually accompanies good
combustion, and for this reason high CO
2 readings alone are not
considered entirely reliable. Wherever an automatic CO
2 recorder is
used, it should be checked from time to time and the analysis carried
further with a view to ascertaining whether there is CO present. As the
percentage of CO
2 in these gases increases, there is a tendency
toward the presence of CO, which, of course, cannot be shown by a CO
2
recorder, and which is often difficult to detect with an Orsat
apparatus. The greatest care should be taken in preparing the cuprous
chloride solution in making analyses and it must be known to be fresh
and capable of absorbing CO.
[Pg 163] In one instance that came to our attention,
in using an Orsat apparatus where the cuprous chloride solution was
believed to be fresh, no CO was indicated in the flue gases but on
passing the same sample into a Hempel apparatus, a considerable
percentage was found. It is not safe, therefore, to assume without
question from a high CO
2 reading that the combustion is
correspondingly good, and the question of excess air alone should be
distinguished from that of good combustion. The effect of a small
quantity of CO, say one per cent, present in the flue gases will have a
negligible influence on the quantity of excess air, but the presence of
such an amount would mean a loss due to the incomplete combustion of the
carbon in the fuel of possibly 4.5 per cent of the total heat in the
fuel burned. When this is considered, the importance of a complete flue
gas analysis is apparent.
Table 34 gives the densities of various gases together with other data
that will be of service in gas analysis work.
TABLE 34
DENSITY OF GASES AT 32 DEGREES FAHRENHEIT AND ATMOSPHERIC PRESSURE
ADAPTED FROM SMITHSONIAN TABLES
| Gas | Chemical
Symbol | Specific Gravity
Air=1 | Weight of
One Cubic Foot
Pounds | Volume of
One Pound
Cubic Feet | Relative Density, Hydrogen = 1 |
|---|
| Exact | Approximate |
|---|
| Oxygen | O | 1.053 | .08922 | 11.208 | 15.87 | 16 |
| Nitrogen | N | 0.9673 | .07829 | 12.773 | 13.92 | 14 |
| Hydrogen | H | 0.0696 | .005621 | 177.90 | 1.00 | 1 |
| Carbon Dioxide | CO2 | 1.5291 | .12269 | 8.151 | 21.83 | 22 |
| Carbon Monoxide | CO | 0.9672 | .07807 | 12.809 | 13.89 | 14 |
| Methane | CH4 | 0.5576 | .04470 | 22.371 | 7.95 | 8 |
| Ethane | C2H6 | 1.075 | .08379 | 11.935 | 14.91 | 15 |
| Acetylene | C2H2 | 0.920 | .07254 | 13.785 | 12.91 | 13 |
| Sulphur Dioxide | SO2 | 2.2639 | .17862 | 5.598 | 31.96 | 32 |
| Air | … | 1.0000 | .08071 | 12.390 | … | …
[Pg 164] [Pl 164] |
|
FOOTNOTES







Alfa Therm Ltd has been specialized in manufacturing top quality Flue Gas Cleaning System with more than 25 years of experience in air pollution control.
ReplyDelete