Greenhouse gas
From Wikipedia, the free encyclopedia
A greenhouse gas (sometimes abbreviated GHG) is a gas in an atmosphere that absorbs and emits radiation within the thermal infrared range. This process is the fundamental cause of the greenhouse effect.[1] The primary greenhouse gases in the Earth's atmosphere are water vapour, carbon dioxide, methane, nitrous oxide, and ozone. In the Solar System, the atmospheres of Venus, Mars, and Titan also contain gases that cause greenhouse effects. Greenhouse gases greatly affect the temperature of the Earth; without them, Earth's surface would be on average about 33 °C (59 °F)[note 1] colder than at present.[2][3][4]However, since the beginning of the Industrial Revolution, the burning of fossil fuels has contributed to the increase in carbon dioxide in the atmosphere from 280 ppm to 390 ppm, despite the uptake of a large portion of the emissions through various natural "sinks" involved in the carbon cycle.[5][6] Anthropogenic carbon dioxide (CO2 ) emissions (i.e., emissions produced by human activities) come from combustion of carbonaceous fuels, principally wood, coal, oil, and natural gas.[7]
Gases in Earth's atmosphere
Greenhouse gases
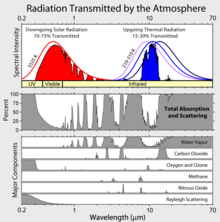
Atmospheric absorption and scattering at different electromagnetic wavelengths. The largest absorption band of carbon dioxide is in the infrared.
- water vapor (H2O)
- carbon dioxide (CO2)
- methane (CH4)
- nitrous oxide (N2O)
- ozone (O3)
Non-greenhouse gases
Although contributing to many other physical and chemical reactions, the major atmospheric constituents, nitrogen (N2), oxygen (O2), and argon (Ar), are not greenhouse gases. This is because molecules containing two atoms of the same element such as N2 and O2 and monatomic molecules such as Argon (Ar) have no net change in their dipole moment when they vibrate and hence are almost totally unaffected by infrared light. Although molecules containing two atoms of different elements such as carbon monoxide (CO) or hydrogen chloride (HCl) absorb IR, these molecules are short-lived in the atmosphere owing to their reactivity and solubility. Because they do not contribute significantly to the greenhouse effect, they are usually omitted when discussing greenhouse gases.Non-greenhouse gases that have an indirect radiative effect
Contribution of clouds to Earth's greenhouse effect
The major non-gas contributor to the Earth's greenhouse effect, clouds, also absorb and emit infrared radiation and thus have an effect on radiative properties of the greenhouse gases. Clouds are water droplets or ice crystals suspended in the atmosphere.[11][12]Impact of a given gas on the overall greenhouse effect
The contribution of each gas to the greenhouse effect is affected by the characteristics of that gas, its abundance, and any indirect effects it may cause. For example, on a molecule-for-molecule basis the direct radiative effects of methane is about 72 times stronger than carbon dioxide over a 20 year time frame[13] but it is present in much smaller concentrations so that its total direct radiative effect is smaller, and it has a shorter atmospheric lifetime. On the other hand, in addition to its direct radiative impact methane has a large indirect radiative effect because it contributes to ozone formation. Shindell et al. (2005)[14] argue that the contribution to climate change from methane is at least double previous estimates as a result of this effect.[15]When these gases are ranked by their direct contribution to the greenhouse effect, the most important are:[11]
| Gas |
Formula |
Contribution (%) |
|---|---|---|
| Water vapor | H2O | 36 – 72 % |
| Carbon dioxide | CO2 | 9 – 26 % |
| Methane | CH4 | 4 – 9 % |
| Ozone | O3 | 3 – 7 % |
Proportion of direct effects at a given moment
It is not possible to state that a certain gas causes an exact percentage of the greenhouse effect. This is because some of the gases absorb and emit radiation at the same frequencies as others, so that the total greenhouse effect is not simply the sum of the influence of each gas. The higher ends of the ranges quoted are for each gas alone; the lower ends account for overlaps with the other gases.[11][12] In addition, some gases such as methane are known to have large indirect effects that are still being quantified.[17]Atmospheric lifetime
Aside from water vapor, which has a residence time of about nine days,[18] major greenhouse gases are well-mixed, and take many years to leave the atmosphere.[19] Although it is not easy to know with precision how long it takes greenhouse gases to leave the atmosphere, there are estimates for the principal greenhouse gases. Jacob (1999)[20] defines the lifetime of an atmospheric species X in a one-box model as the average time that a molecule of X remains in the box. Mathematically
of an atmospheric species X in a one-box model as the average time that a molecule of X remains in the box. Mathematically  can be defined as the ratio of the mass
can be defined as the ratio of the mass 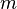 (in kg) of X in the box to its removal rate, which is the sum of the flow of X out of the box (
(in kg) of X in the box to its removal rate, which is the sum of the flow of X out of the box ( ), chemical loss of X (
), chemical loss of X (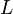 ), and deposition of X (
), and deposition of X (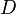 ) (all in kg/sec):
) (all in kg/sec):  .[20] If one stopped pouring any of this gas into the box, then after a time
.[20] If one stopped pouring any of this gas into the box, then after a time  , its concentration would be about halved.
, its concentration would be about halved.The atmospheric lifetime of a species therefore measures the time required to restore equilibrium following a sudden increase or decrease in its concentration in the atmosphere. Individual atoms or molecules may be lost or deposited to sinks such as the soil, the oceans and other waters, or vegetation and other biological systems, reducing the excess to background concentrations. The average time taken to achieve this is the mean lifetime.
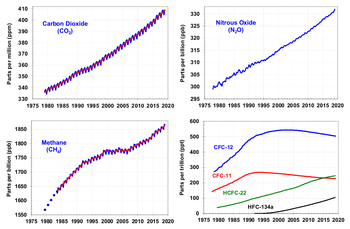
Carbon dioxide has a variable atmospheric lifetime, and cannot be specified precisely.[21] The atmospheric lifetime of CO2 is estimated of the order of 30–95 years.[22] This figure accounts for CO2 molecules being removed from the atmosphere by mixing into the ocean, photosynthesis, and a few other processes. However, this excludes the balancing fluxes of CO2 into the atmosphere from the geological reservoirs, which have slower characteristic rates.[23] While more than half of the CO2 emitted is currently removed from the atmosphere within a century, some fraction (about 20%) of emitted CO2 remains in the atmosphere for many thousands of years.[24][25][26]
Global warming potential
The global warming potential (GWP) depends on both the efficiency of the molecule as a greenhouse gas and its atmospheric lifetime. GWP is measured relative to the same mass of CO2 and evaluated for a specific timescale. Thus, if a gas has a high radiative forcing but also a short lifetime, it will have a large GWP on a 20 year scale but a small one on a 100 year scale. Conversely, if a molecule has a longer atmospheric lifetime than CO2 its GWP will increase with the timescale considered. Carbon dioxide is defined to have a GWP of 1 over all time periods.Methane has an atmospheric lifetime of 12 ± 3 years and a GWP of 72 over 20 years, 25 over 100 years and 7.6 over 500 years. The decrease in GWP at longer times is because methane is degraded to water and CO2 through chemical reactions in the atmosphere.
Examples of the atmospheric lifetime and GWP relative to CO2 for several greenhouse gases are given in the following table:[13]
| Gas name | Chemical formula |
Lifetime (years) |
Global warming potential (GWP) for given time horizon | ||
|---|---|---|---|---|---|
| 20-yr | 100-yr | 500-yr | |||
| Carbon dioxide | CO2 | See above | 1 | 1 | 1 |
| Methane | CH4 | 12 | 72 | 25 | 7.6 |
| Nitrous oxide | N2O | 114 | 289 | 298 | 153 |
| CFC-12 | CCl2F2 | 100 | 11 000 | 10 900 | 5 200 |
| HCFC-22 | CHClF2 | 12 | 5 160 | 1 810 | 549 |
| Tetrafluoromethane | CF4 | 50 000 | 5 210 | 7 390 | 11 200 |
| Hexafluoroethane | C2F6 | 10 000 | 8 630 | 12 200 | 18 200 |
| Sulphur hexafluoride | SF6 | 3 200 | 16 300 | 22 800 | 32 600 |
| Nitrogen trifluoride | NF3 | 740 | 12 300 | 17 200 | 20 700 |
Natural and anthropogenic sources
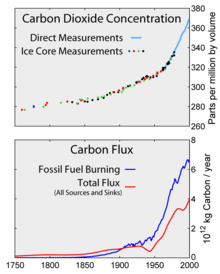
Top: Increasing atmospheric carbon dioxide levels as measured in the atmosphere and reflected in ice cores. Bottom: The amount of net carbon increase in the atmosphere, compared to carbon emissions from burning fossil fuel.
The 2007 Fourth Assessment Report compiled by the IPCC (AR4) noted that "changes in atmospheric concentrations of greenhouse gases and aerosols, land cover and solar radiation alter the energy balance of the climate system", and concluded that "increases in anthropogenic greenhouse gas concentrations is very likely to have caused most of the increases in global average temperatures since the mid-20th century".[31] In AR4, "most of" is defined as more than 50%.
| Gas | Preindustrial level | Current level | Increase since 1750 | Radiative forcing (W/m2) |
|---|---|---|---|---|
| Carbon dioxide | 280 ppm | 396 ppm | 116 ppm | 1.46 |
| Methane | 700 ppb | 1745 ppb | 1045 ppb | 0.48 |
| Nitrous oxide | 270 ppb | 314 ppb | 44 ppb | 0.15 |
| CFC-12 | 0 | 533 ppt | 533 ppt | 0.17 |
Anthropogenic greenhouse gases
It is likely that anthropogenic warming, such as that due to elevated greenhouse gas levels, has had a discernible influence on many physical and biological systems. Warming is projected to affect various issues such as freshwater resources, industry, food and health.[42]
The main sources of greenhouse gases due to human activity are:
- burning of fossil fuels and deforestation leading to higher carbon dioxide concentrations in the air. Land use change (mainly deforestation in the tropics) account for up to one third of total anthropogenic CO2 emissions.[41]
- livestock enteric fermentation and manure management,[43] paddy rice farming, land use and wetland changes, pipeline losses, and covered vented landfill emissions leading to higher methane atmospheric concentrations. Many of the newer style fully vented septic systems that enhance and target the fermentation process also are sources of atmospheric methane.
- use of chlorofluorocarbons (CFCs) in refrigeration systems, and use of CFCs and halons in fire suppression systems and manufacturing processes.
- agricultural activities, including the use of fertilizers, that lead to higher nitrous oxide (N2O) concentrations.
| Seven main fossil fuel combustion sources |
Contribution (%) |
|---|---|
| Liquid fuels (e.g., gasoline, fuel oil) | 36 % |
| Solid fuels (e.g., coal) | 35 % |
| Gaseous fuels (e.g., natural gas) | 20 % |
| Cement production | 3 % |
| Flaring gas industrially and at wells | < 1 % |
| Non-fuel hydrocarbons | < 1 % |
| "International bunker fuels" of transport not included in national inventories |
4 % |
Carbon dioxide, methane, nitrous oxide and three groups of fluorinated gases (sulfur hexafluoride, HFCs, and PFCs) are the major greenhouse gases and the subject of the Kyoto Protocol, which came into force in 2005.[46]
Although CFCs are greenhouse gases, they are regulated by the Montreal Protocol, which was motivated by CFCs' contribution to ozone depletion rather than by their contribution to global warming. Note that ozone depletion has only a minor role in greenhouse warming though the two processes often are confused in the media.
On December 7, 2009, the US Environmental Protection Agency released its final findings on greenhouse gases, declaring that "greenhouse gases (GHGs) threaten the public health and welfare of the American people". The finding applied to the same "six key well-mixed greenhouse gases" named in the Kyoto Protocol: carbon dioxide, methane, nitrous oxide, hydrofluorocarbons, perfluorocarbons, and sulfur hexafluoride.[47][48]
Role of water vapor
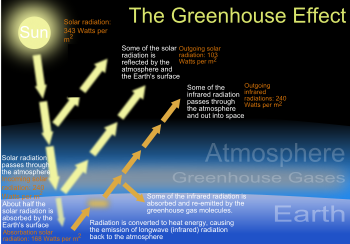
Water vapor accounts for the largest percentage of the greenhouse effect, between 36% and 66% for clear sky conditions and between 66% and 85% when including clouds.[12] Water vapor concentrations fluctuate regionally, but human activity does not significantly affect water vapor concentrations except at local scales, such as near irrigated fields. The atmospheric concentration of vapor is highly variable, from less than 0.01% in extremely cold regions up to 20% in warm, humid regions.[49]The average residence time of a water molecule in the atmosphere is only about nine days, compared to years or centuries for other greenhouse gases such as CH4 and CO2. Thus, water vapor responds to and amplifies effects of the other greenhouse gases. The Clausius-Clapeyron relation establishes that air can hold more water vapor per unit volume when it warms. This and other basic principles indicate that warming associated with increased concentrations of the other greenhouse gases also will increase the concentration of water vapor. Because water vapor is a greenhouse gas, this results in further warming and so is a "positive feedback" that amplifies the original warming. Eventually other earth processes offset these positive feedbacks, stabilizing the global temperature at a new equilibrium and preventing the loss of Earth's water through a Venus-like runaway greenhouse effect.[50]
Atmospheric concentration
Measurements from Antarctic ice cores show that before industrial emissions started atmospheric CO2 mole fractions were about 280 parts per million (ppm), and stayed between 260 and 280 during the preceding ten thousand years.[51] Carbon dioxide mole fractions in the atmosphere have gone up by approximately 35 percent since the 1900s, rising from 280 parts per million by volume to 387 parts per million in 2009. One study using evidence from stomata of fossilized leaves suggests greater variability, with carbon dioxide mole fractions above 300 ppm during the period seven to ten thousand years ago,[52] though others have argued that these findings more likely reflect calibration or contamination problems rather than actual CO2 variability.[53][54] Because of the way air is trapped in ice (pores in the ice close off slowly to form bubbles deep within the firn) and the time period represented in each ice sample analyzed, these figures represent averages of atmospheric concentrations of up to a few centuries rather than annual or decadal levels.Since the beginning of the Industrial Revolution, the concentrations of most of the greenhouse gases have increased. For example, the mole fraction of carbon dioxide has increased from 280 ppm by about 36% to 380 ppm, or 100 ppm over modern pre-industrial levels. The first 50 ppm increase took place in about 200 years, from the start of the Industrial Revolution to around 1973; however the next 50 ppm increase took place in about 33 years, from 1973 to 2006.[55]
Recent data also shows that the concentration is increasing at a higher rate. In the 1960s, the average annual increase was only 37% of what it was in 2000 through 2007.[56]
Today, the stock of carbon in the atmosphere increases by more than 3 million tonnes per annum (0.04%) compared with the existing stock.[clarification needed] This increase is the result of human activities by burning fossil fuels, deforestation and forest degradation in tropical and boreal regions.[57]
The other greenhouse gases produced from human activity show similar increases in both amount and rate of increase. Many observations are available online in a variety of Atmospheric Chemistry Observational Databases.
| Gas | Current (1998) Amount by volume |
Increase (absolute, ppm) over pre-industrial (1750) |
Increase (relative, %) over pre-industrial (1750) |
Radiative forcing (W/m2) |
|---|---|---|---|---|
| Carbon dioxide | 365 ppm (383 ppm, 2007.01) |
87 ppm (105 ppm, 2007.01) |
31 % (38 %, 2007.01) |
1.46 (~1.53, 2007.01) |
| Methane | 1745 ppb | 1045 ppb | 150 % | 0.48 |
| Nitrous oxide | 314 ppb | 44 ppb | 16 % | 0.15 |
| Gas | Current (1998) Amount by volume |
Radiative forcing (W/m2) |
|---|---|---|
| CFC-11 | 268 ppt | 0.07 |
| CFC-12 | 533 ppt | 0.17 |
| CFC-113 | 84 ppt | 0.03 |
| Carbon tetrachloride | 102 ppt | 0.01 |
| HCFC-22 | 69 ppt | 0.03 |
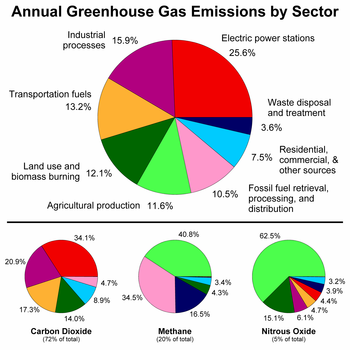
Greenhouse gas emissions ("sources")
Between the period 1970 to 2004, GHG emissions (measured in CO2-equivalent)[60] increased at an average rate of 1.6% per year, with CO2 emissions from the use of fossil fuels growing at a rate of 1.9% per year.[61][62] Total anthropogenic emissions at the end of 2009 were estimated at 49.5 gigatonnes CO2-equivalent.[63] These emissions include CO2 from fossil fuel use and from land use, as well as emissions of methane, nitrous oxide and other GHGs covered by the Kyoto Protocol.At present, the two primary sources of CO2 emissions are from burning coal used for electricity generation and petroleum used for motor transport.[citation needed]
Regional and national attribution of emissions
See also: Kyoto Protocol and government action
There are several different ways of measuring GHG emissions (see
World Bank (2010, p. 362) for a table of national emissions data).[64]Some variables that have been reported[65] include:
- Definition of measurement boundaries. Emissions can be attributed geographically, to the area where they were emitted (the territory principle) or by the activity principle to the territory that caused the emissions to be produced. These two principles would result in different totals when measuring for example the importation of electricity from one country to another or the emissions at an international airport.
- The time horizon of different GHGs. Contribution of a given GHG is reported as a CO2 equivalent; the calculation to determine this takes into account how long that gas remains in the atmosphere. This is not always known accurately and calculations must be regularly updated to take into account new information.
- What sectors are included in the calculation (e.g. energy industries, industrial processes, agriculture etc.). There is often a conflict between transparency and availability of data.
- The measurement protocol itself. This may be via direct measurement or estimation; the four main methods are the emission factor-based method, the mass balance method, the predictive emissions monitoring system and the continuing emissions monitoring systems. The methods differ in accuracy, but also in cost and usability.
Emissions may be measured over long time periods. This measurement type is called historical or cumulative emissions. Cumulative emissions give some indication of who is responsible for the build-up in the atmospheric concentration of GHGs (IEA, 2007, p. 199).[67]
The national accounts balance would be positively related to carbon emissions. The national accounts balance shows the difference between exports and imports. For many richer nations, such as the United States, the accounts balance is negative because more goods are imported than they are exported. This is mostly due to the fact that it is cheaper to produce goods outside of developed countries, leading the economies of developed countries to become increasingly dependent on services and not goods. We believed that a positive accounts balance would means that more production was occurring in a country, so more factories working would increase carbon emission levels.(Holtz-Eakin, 1995, pp.;85;101).[68]
Emissions may also be measured across shorter time periods. Emissions changes may, for example, be measured against a base year of 1990. 1990 was used in the United Nations Framework Convention on Climate Change (UNFCCC) as the base year for emissions, and is also used in the Kyoto Protocol (some gases are also measured from the year 1995) (Grubb, 2003, pp. 146, 149).[69] A country's emissions may also be reported as a proportion of global emissions for a particular year.
Another measurement is of per capita emissions. This divides a country's total annual emissions by its mid-year population (World Bank, 2010, p. 370). Per capita emissions may be based on historical or annual emissions (Banuri et al., 1996, pp. 106–107).
Greenhouse gas intensity and land-use change
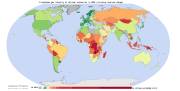
Map of cumulative per capita anthropogenic atmospheric CO2 emissions by country. Cumulative emissions include land use change, and are measured between the years 1950 and 2000.
Land-use change, e.g., the clearing of forests for agricultural use, can affect the concentration of GHGs in the atmosphere by altering how much carbon flows out of the atmosphere into carbon sinks.[71] Accounting for land-use change can be understood as an attempt to measure “net” emissions, i.e., gross emissions from all GHG sources minus the removal of emissions from the atmosphere by carbon sinks (Banuri et al., 1996, pp. 92–93).
There are substantial uncertainties in the measurement of net carbon emissions.[72] Additionally, there is controversy over how carbon sinks should be allocated between different regions and over time (Banuri et al., 1996, p. 93). For instance, concentrating on more recent changes in carbon sinks is likely to favour those regions that have deforested earlier, e.g., Europe.
Cumulative and historical emissions
Cumulative anthropogenic (i.e., human-emitted) emissions of CO2 from fossil fuel use are a major cause of global warming,[73] and give some indication of which countries have contributed most to human-induced climate change.[74]:15| Region | Industrial CO2 |
Total CO2 |
|---|---|---|
| OECD North America | 33.2 | 29.7 |
| OECD Europe | 26.1 | 16.6 |
| Former USSR | 14.1 | 12.5 |
| China | 5.5 | 6.0 |
| Eastern Europe | 5.5 | 4.8 |
Including biotic emissions brings about the same controversy mentioned earlier regarding carbon sinks and land-use change (Banuri et al., 1996, pp. 93–94). The actual calculation of net emissions is very complex, and is affected by how carbon sinks are allocated between regions and the dynamics of the climate system.
Non-OECD countries accounted for 42% of cumulative energy-related CO2 emissions between 1890-2007.[75]:179-180 Over this time period, the US accounted for 28% of emissions; the EU, 23%; Russia, 11%; China, 9%; other OECD countries, 5%; Japan, 4%; India, 3%; and the rest of the world, 18%.[75]:179-180
Changes since a particular base year
Between 1970-2004, global growth in annual CO2 emissions was driven by North America, Asia, and the Middle East.[76] The sharp acceleration in CO2 emissions since 2000 to more than a 3% increase per year (more than 2 ppm per year) from 1.1% per year during the 1990s is attributable to the lapse of formerly declining trends in carbon intensity of both developing and developed nations. China was responsible for most of global growth in emissions during this period. Localised plummeting emissions associated with the collapse of the Soviet Union have been followed by slow emissions growth in this region due to more efficient energy use, made necessary by the increasing proportion of it that is exported.[44] In comparison, methane has not increased appreciably, and N2O by 0.25% y−1.Using different base years for measuring emissions has an effect on estimates of national contributions to global warming.[74]:17-18[77] This can be calculated by dividing a country's highest contribution to global warming starting from a particular base year, by that country's minimum contribution to global warming starting from a particular base year. Choosing between different base years of 1750, 1900, 1950, and 1990 has a significant effect for most countries.[74]:17-18 Within the G8 group of countries, it is most significant for the UK, France and Germany. These countries have a long history of CO2 emissions (see the section on Cumulative and historical emissions).
Annual emissions
Annual per capita emissions in the industrialized countries are typically as much as ten times the average in developing countries.[69]:144 Due to China's fast economic development, its annual per capita emissions are quickly approaching the levels of those in the Annex I group of the Kyoto Protocol (i.e., the developed countries excluding the USA).[78] Other countries with fast growing emissions are South Korea, Iran, and Australia. On the other hand, annual per capita emissions of the EU-15 and the USA are gradually decreasing over time.[78] Emissions in Russia and the Ukraine have decreased fastest since 1990 due to economic restructuring in these countries.[79]Energy statistics for fast growing economies are less accurate than those for the industrialized countries. For China's annual emissions in 2008, the Netherlands Environmental Assessment Agency estimated an uncertainty range of about 10%.[78]
Top emitters
See also: List of countries by carbon dioxide emissions, List of countries by carbon dioxide emissions per capita, List of countries by greenhouse gas emissions, and List of countries by greenhouse gas emissions per capita
Annual
In 2009, the annual top ten emitting countries accounted for about two-thirds of the world's annual energy-related CO2 emissions.[80]| Country | % of global total annual emissions |
Tonnes of GHG per capita |
|---|---|---|
| People's Rep. of China | 23.6 | 5.13 |
| United States | 17.9 | 16.9 |
| India | 5.5 | 1.37 |
| Russian Federation | 5.3 | 10.8 |
| Japan | 3.8 | 8.6 |
| Germany | 2.6 | 9.2 |
| Islamic Rep. of Iran | 1.8 | 7.3 |
| Canada | 1.8 | 15.4 |
| Korea | 1.8 | 10.6 |
| United Kingdom | 1.6 | 7.5 |
Cumulative
| Country | % of world total |
Metric tonnes CO2 per person |
|---|---|---|
| United States | 28.5 | 1,132.7 |
| China | 9.36 | 85.4 |
| Russian Federation | 7.95 | 677.2 |
| Germany | 6.78 | 998.9 |
| United Kingdom | 5.73 | 1,127.8 |
| Japan | 3.88 | 367 |
| France | 2.73 | 514.9 |
| India | 2.52 | 26.7 |
| Canada | 2.17 | 789.2 |
| Ukraine | 2.13 | 556.4 |
Embedded emissions
One way of attributing greenhouse gas (GHG) emissions is to measure the embedded emissions (also referred to as "embodied emissions") of goods that are being consumed. Emissions are usually measured according to production, rather than consumption.[83] For example, in the main international treaty on climate change (the UNFCCC), countries report on emissions produced within their borders, e.g., the emissions produced from burning fossil fuels.[75]:179[84]:1 Under a production-based accounting of emissions, embedded emissions on imported goods are attributed to the exporting, rather than the importing, country. Under a consumption-based accounting of emissions, embedded emissions on imported goods are attributed to the importing country, rather than the exporting, country.Davis and Caldeira (2010)[84]:4 found that a substantial proportion of CO2 emissions are traded internationally. The net effect of trade was to export emissions from China and other emerging markets to consumers in the US, Japan, and Western Europe. Based on annual emissions data from the year 2004, and on a per-capita consumption basis, the top-5 emitting countries were found to be (in tCO2 per person, per year): Luxembourg (34.7), the US (22.0), Singapore (20.2), Australia (16.7), and Canada (16.6).[84]:5
Effect of policy
Rogner et al. (2007) assessed the effectiveness of policies to reduce emissions (mitigation of climate change).[61] They concluded that mitigation policies undertaken by UNFCCC Parties were inadequate to reverse the trend of increasing GHG emissions. The impacts of population growth, economic development, technological investment, and consumption had overwhelmed improvements in energy intensities and efforts to decarbonize (energy intensity is a country's total primary energy supply (TPES) per unit of GDP (Rogner et al., 2007).[85] TPES is a measure of commercial energy consumption (World Bank, 2010, p. 371)).[64]Projections
Based on then-current energy policies, Rogner et al. (2007) projected that energy-related CO2 emissions in 2030 would be 40-110% higher than in 2000.[61] Two-thirds of this increase was projected to come from non-Annex I countries. Per capita emissions in Annex I countries were still projected to remain substantially higher than per capita emissions in non-Annex I countries. Projections consistently showed a 25-90% increase in the Kyoto gases (carbon dioxide, methane, nitrous oxide, sulphur hexafluoride) compared to 2000.IEA (2007, p. 199) estimated future cumulative energy-related CO2 emissions for several countries.[67] Their reference scenario projected cumulative energy-related CO2 emissions between the years 1900 and 2030. In this scenario, China’s share of cumulative emissions rises to 16%, approaching that of the United States (25%) and the European Union (18%). India’s cumulative emissions (4%) approach those of Japan (4%).
Relative CO2 emission from various fuels
One liter of gasoline, when used as a fuel, produces 2.32 kg (1.3 cubic meters) of carbon dioxide, a greenhouse gas. One US gallon produces 19.4 lb (172.65 cubic feet)[86][87][88]| Fuel name | CO2 emitted (lbs/106 Btu) |
CO2 emitted (g/106 J) |
|---|---|---|
| Natural gas | 117 | 50.30 |
| Liquefied petroleum gas | 139 | 59.76 |
| Propane | 139 | 59.76 |
| Aviation gasoline | 153 | 65.78 |
| Automobile gasoline | 156 | 67.07 |
| Kerosene | 159 | 68.36 |
| Fuel oil | 161 | 69.22 |
| Tires/tire derived fuel | 189 | 81.26 |
| Wood and wood waste | 195 | 83.83 |
| Coal (bituminous) | 205 | 88.13 |
| Coal (sub-bituminous) | 213 | 91.57 |
| Coal (lignite) | 215 | 92.43 |
| Petroleum coke | 225 | 96.73 |
| Coal (anthracite) | 227 | 97.59 |











No comments:
Post a Comment